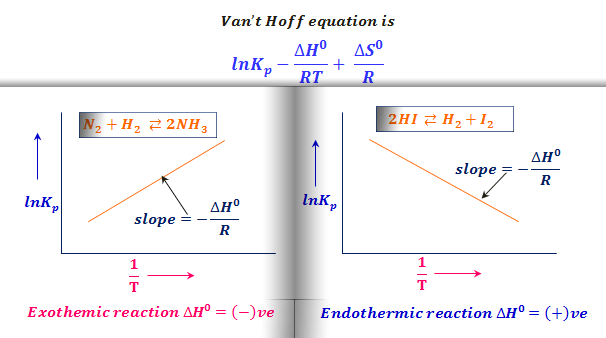
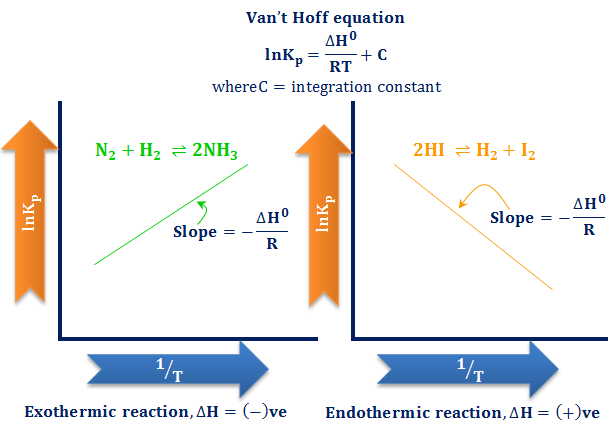
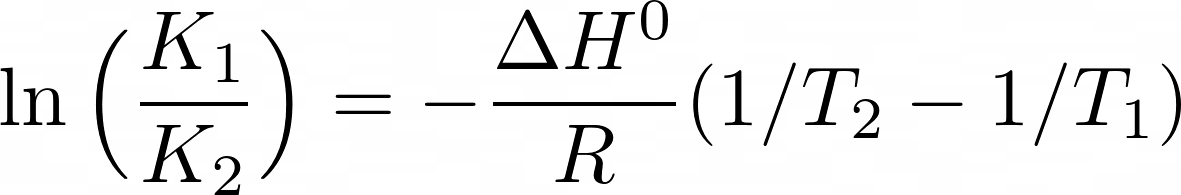ANALOGY BETWEEN ARRHENIUS AND VAN'T HOFF EQUATION :— {Topic: Chemical Kinetics} Arrhenius equation and Van't Hoff equation are two important and often asked about equations in chemistry. Remembering one of these can

Jacobus Henricus van 't Hoff - Integrated van't Hoff equation (if the standard reaction enthalpy ΔH0 is known and constant in the temperature range of the measurement) is used for temperature correction.

CHE102 - Lesson 19-van't Hoff Equation - Vant Hoff Equation The Vant Hoff Equation Can Be Used To Estimate Kc Or Kp At A Given Temperature Provided That Kc Or | Course Hero

SOLVED: TC = g C o RT van't Hoff equation Osmotic pressure (atm) of particles per mole solution (Osm mol) Concentration(mmol Reflection coefficient (vares from Gas constant 082 atm mol Absolute temperature (

PDF) Van't Hoff equation-equilibrium How much Van't Hoff equation -effect on temperature | Kajal Panda - Academia.edu